|
Because it contains three medicines, it may be useful for those on a fixed-dose combination of fluticasone / vilanterol (Breo Ellipta) who need added bronchodilation, or those who are already using Breo Ellipta and Incruse Ellipta (umeclidinium). Trelegy Ellipta (fluticasone furoate / umeclidinium / vilanterol) contains three medications in one inhaler for the treatment of adults with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and emphysema, and for the treatment of asthma. In September 2020, the FDA approved a third indication for Trelegy for maintenance treatment of asthma in adults. In April 2018, the indications were expanded to include the daily maintenance treatment of airflow obstruction in patients with COPD. In September 2017, the FDA approved Trelegy Ellipta for the long-term maintenance of chronic obstructive pulmonary disease (COPD). If you do not have a fast-acting rescue inhaler, contact your health care provider to have one prescribed for you. 
Use only a fast-acting inhalation medicine (for example, albuterol) for an attack. It does not work fast enough to treat a bronchospasm attack. Trelegy Ellipta is not a fast-acting rescue medicine to be used during an asthma attack. 
It combines an inhaled corticosteroid (ICS), a long-acting muscarinic antagonist (LAMA) and long-acting beta2-adrenergic agonist (LABA). Trelegy Ellipta is a once-daily therapy that combines three medicines into one inhaler. 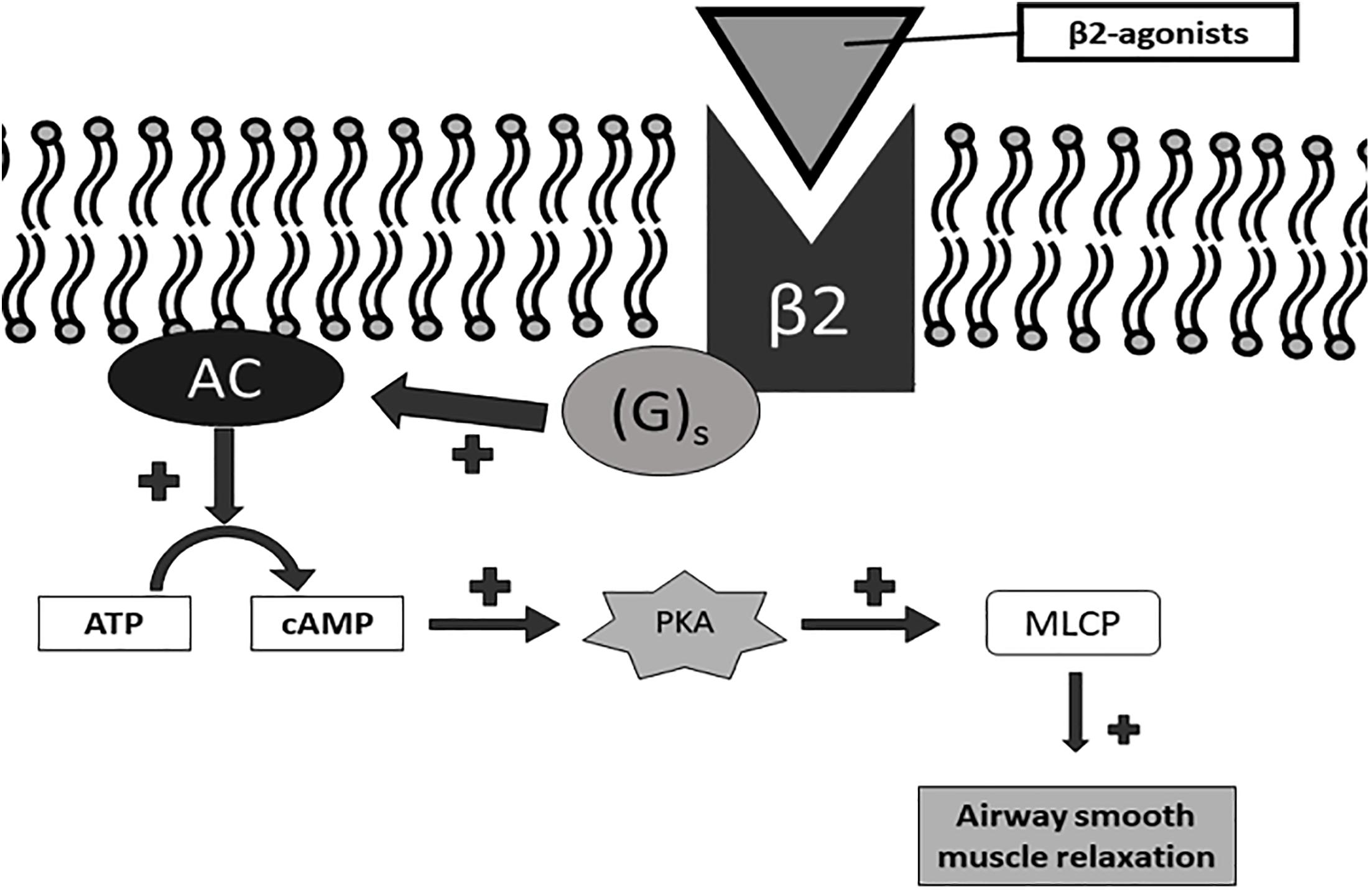
Trelegy Ellipta is not used as a fast-acting (rescue) inhaler for the relief of acute bronchospasm.Trelegy Ellipta is also approved for use in adults with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and emphysema.Yes, Trelegy Ellipta (generic name: fluticasone furoate / umeclidinium / vilanterol) is now approved for the maintenance treatment of asthma in patients aged 18 years and older.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
